Citation: Kraus R, “Trends in Anti-Glaucoma Topical Treatments”. ONdrugDelivery Magazine, Issue 94 (Jan 2019), pp 40-42.
Rouven Kraus presents the case for multidose delivery systems rather than single-use disposables, and introduces Aero Pump’s Ophthalmic Multidose System which utilises the company’s proprietary 3K®-technology for preservative-free, multidose eyedroppers.
“The launch of novel anti-glaucoma drugs with additional neuroprotective properties that avoid loss of retinal ganglion cells is set to become a major breakthrough in glaucoma treatment…”
The ophthalmic drug market is one of the fastest growing market sectors in the pharmaceutical industry. More than 285 million people suffer from visual impairments worldwide, a number expected to rise due to the increasing prevalence of ophthalmic disorders. Such disorders are on the rise primarily because of extended life expectancy in developed countries (ageing populations), increased air pollution and higher numbers of patients being diagnosed with diabetes.

Figure 1: Thea Pharma’s Duokopt containing dorzolamide and timolol in the 3K®-system and the ergonomically designed Easygrip®, which enables a safe grip and an easy administration of the drops for the patient.
A total of 962 million people globally were aged 65 years or over in 2017, however, the number of people in this age bracket is expected to nearly double by 2050 – a total of 1.6 billion people. 5% of people aged 65 or over develop glaucoma, a group of optic neuropathies resulting from damage to the optic nerve caused by intraocular pressure. Glaucoma is the second leading cause of irreversible blindness. With a predicted 80 million people affected by glaucoma by 2020, it will be one of the most prevalent ocular diseases.
Drugs are usually the first line of treatment for primary open-angle glaucoma (POAG), the most common type of glaucoma. Alpha agonists like brimonidine decrease the production of aqueous humour and increase uveoscleral outflow. Beta blockers (e.g. timolol) and carbonic anhydrase inhibitors (e.g. dorzolamide, brinzolamide) decrease the aqueous humour through the ciliary body, whereas miotics (e.g. pilocarpine) and prostaglandin analogues (e.g. latanoprost, bimatoprost, travoprost) improve the drainage. Rho khinase inhibitors (e.g. netarsudil) are a new class of glaucoma drugs, first launched in 2018. Rho khinase inhibitor eyedrops reduce the elevated intraocular pressure (IOP) by suppressing the rho khinase enzymes that produce the aqueous humour.
Of these, prostaglandin analogues are the standard first-line treatment to decrease IOP due to their advantage of greater efficacy. Manufacturers have launched combination products that combine prostaglandin analogues with a beta-blocker, which are predominantly to be used once or twice a day.

Figure 2: Bausch + Lomb’s Vizitrav® containing Travoprost in the 3K®-system.
Single-use vials (often produced in the blow-fill-seal technology) do not protect the contents from contamination after they are opened, meaning the patient needs to discard them after the administration of just one dose. Patients often do not follow those instructions and use their single-use vials more than once. As such, the superior option would be to use multidose containers, which enable repeated use of the product to deliver the required dose daily.
The industry has been challenged to design multidose eyedroppers which satisfy all the regulatory guidelines set out by the authorities. The EMA recommends sterile eyedrops to be preservative-free, as it is known that preserving agents cause side-effects like irritation of the ocular surface, and to dispense a defined metered dose with each drop.
Preservative-free, multidose systems either have an airless container like the COMOD®-system or require a special sinter filter with antimicrobial properties to compensate for the vacuum formed inside the container after the drop is dispensed, as well as a valve that forms a microbiologically tight seal (e.g. the 3K®-technology in Aero Pump’s Ophthalmic Multidose System).
“Aero Pump GmbH, together with its partner URSATEC Verpackung GmbH, have developed a preservative-free, multidose system with their proprietary 3K®-technology…”
Due to the expiry of patents relating to anti-glaucoma products in the past few years, many generic products with a prostaglandine analogue are currently in development. Based on the latest guidelines, those manufacturers are often looking to launch their products in preservative-free, multidose containers.
The launch of novel anti-glaucoma drugs with additional neuroprotective properties that avoid loss of retinal ganglion cells is set to become a major breakthrough in glaucoma treatment. Currently, numerous clinical trials are taking place, combining formulations with the benefits of using just one bottle for several medications. For glaucoma patients and ophthalmologists, it is an exciting time as these new medications promise improved features by directly targeting the trabecular meshwork.
AERO PUMP’S PRESERVATIVE-FREE OPHTHALMIC MULTIDOSE SYSTEM
“The actuation force of Aero Pump’s Ophthalmic Multidose System is stable and independent of the residual liquid inside the container. This fact is especially noticeable for older patients where it can be very difficult to eject the last few drops of the liquid…”
Aero Pump GmbH, together with its partner URSATEC Verpackung GmbH, have developed a preservative-free, multidose system with their proprietary 3K®-technology (Figures 1 and 2). Special germ-reducing components inside the 3K®-system ensure the microbiological integrity and safety of the device. This pump system is available for use with plastic or glass containers and, in terms of reducing container interaction with the product, this is a particular advantage.
The 3K®-system delivers an accurate dose across the full lifecycle of the product, with one measured drop per actuation. Conventional squeeze-actuated devices on the other hand are known to have an imprecise dose accuracy, some can even create an extremely uncomfortable jet when squeezed.

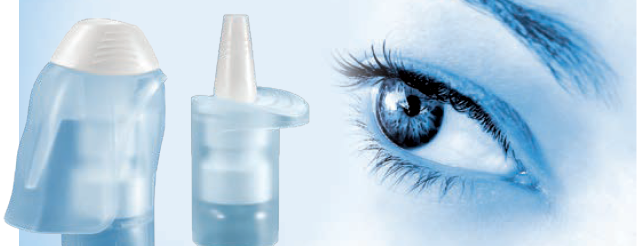
Figure 3: The Ophthalmic Multidose System in different sleeve designs.
The actuation force of Aero Pump’s Ophthalmic Multidose System is stable and independent of the residual liquid inside the container. This fact is especially noticeable for older patients where it can be very difficult to eject the last few drops of the liquid, which can often lead to an increased residual volume when squeeze-actuated devices are used.
Alongside the development of ophthalmic multidose devices, Aero Pump has developed various customer-friendly actuation aids that enable a convenient application of the drop into the eye of the patient (Figure 3).