Citation: Dessertenne E, “Connected Devices: the Next Step in Healthcare Innovation”. ONdrugDelivery Magazine, Issue 92 (Dec 2018), pp 16-19.
Eric Dessertenne discusses the current status of connectivity in healthcare, its future, benefits both in terms of addressing adherence and big data analysis, and emphasises the need for a robust business case. He goes on to describe Biocorp’s portfolio of connected delivery systems, including both integrated devices and connectivity add-ons.
INTRODUCTION
“Adherence is not the sole reason behind the rise of connectivity. Today’s world is ravenous for data and the insights that can be gleaned from its analysis…”
We live in an ever more connected world; a fact clear even to the most casual observer. Across industries there is a drive to include smart and connected features into new technology developments to meet a demand for convenience and advanced capabilities from consumers, and for data to analyse from business. Even in the conservative world of pharma and healthcare, where the well-tested and thoroughly understood often seems preferable to cutting-edge innovation in the face of strict regulatory bodies, the drive towards connectivity is inexorable and undeniable.
A recent article in Health Data Management1 discussed predictions for the coming years in connected healthcare. Amongst them it suggested that, by 2019, more than 50% of life science and healthcare firms will be utilising real-world evidence and, amongst those companies, digital mobile engagement will have increased by 50%; by 2020, adoption of Internet of Things (IoT) based assets in hospitals will have doubled, and 25% of data used in medical care will be captured by the patient themselves; and by 2021 digital healthcare services will account for 6% of all global healthcare expenditure.
In today’s connected world, the consumer tools of the “wellbeing” industry are being converted into actual healthcare products, in some cases by a natural progression of businesses such as Fitbit, in others by patients making their own “home-made” digital healthcare solutions. Regulators have begun the process of establishing their position, and clinical outcomes for connected devices are starting to arrive. For example, in April 2017, Merck Serono reported results from interferon beta-1a delivered by the RebiSmart™ connected device, which not only achieved a very positive impact on adherence (>95% adherence rate) but, crucially, increased the relapse-free rate to more than 77% over 140 weeks, establishing a clear link between better adherence and lower relapse rate.
“Biocorp has long understood that connectivity in drug delivery devices needs to be considered carefully, designed for a specific usage and presented with a robust business case…”
The underlying factors of the buzz around connectivity in healthcare are well documented. Most often cited, as with the RebiSmart™, is the promise connectivity presents to tackling the adherence challenge, a huge drain on healthcare both in terms of patients failing to reach their desired therapeutic outcomes and in financial cost to healthcare institutions and the pharma industry itself. Connected devices, with the ability to provide patients with calendars, reminders and feedback on their own treatment, offer one of the most compelling answers to this challenge. As such, device designers across the industry have thrown themselves into the task of bringing connected devices to market.
Of course, adherence is not the sole reason behind the rise of connectivity. Today’s world is ravenous for data and the insights that can be gleaned from its analysis. Pharma is eager to realise the benefits this presents in healthcare, including gaining insights into real-world use of medication and devices to feed back into future designs, outcome-based payment models, and IoT-integrated manufacturing and logistics to smooth and enhance production and delivery chains.
Connectivity applied in the context of the current healthcare goal of moving treatment from the clinic to the home is a clear winner. This is evidenced by the success of Abbott’s FreeStyle Libre blood glucose monitoring (BGM) device, which greatly increased ease-of-use for patients compared with previous BGM technologies and rapidly proved its worth to payers after a successful launch in Europe.
Biocorp has been working in the area of connectivity since 2013. It has long understood that connectivity in drug delivery devices needs to be considered carefully, designed for a specific usage and presented with a robust business case. As such, Biocorp has developed a portfolio of connected devices in both add-on and integrated formats, spanning both parenteral and respiratory drug delivery.
SOLVING CONNECTED CARE FOR DIABETES

Figure 1: Easylog, smart cap for pen injectors.
Based on the observation that some critical needs of diabetic patients are not being fulfilled, Biocorp has developed Easylog™ (Figure 1) is a connected smart cap that is compatible with all pen injectors, which records and logs the exact dose dialled and injected by the patient, along with the time and date. The data is sent to a mobile app using Bluetooth technology (Figure 2).
Easylog is an add-on device, meaning it can be used with existing products without designing a new product from the ground up and going through a full combination product approval process. The add-on approach enables connectivity to be readily deployed, in effect allowing pharma companies to gain real experience with a connected device in their product portfolio, without any impact on existing industrial processes and infrastructure.

Figure 2: Easylog records injected dose and time, transfers it via Bluetooth to a secure server and outputs useful data to patients and healthcare providers.
The pen injector market contains a lot of disposable products. By using an add-on such as Easylog, the cost of adding connectivity to a device can be offset to the reusable add-on. Easylog is reusable for up to two years. Disposable pen injectors represent a major portion of the market, especially when looking at diabetes, which itself is the largest market in this area by volume.
The diabetes market is, on the whole, already very well connected when looking at BGMs and other monitoring devices. By bringing the pen injector into the connected sphere the missing piece of the puzzle can be filled in, and a comprehensive connected approach to diabetes care can be offered. This is a clear unmet need in the diabetes market and Easylog is in prime position to meet it by adding the recorded insulin injection time and dosage data to the patient’s logs, enabling them to accurately track their insulin usage and, in tandem with a connected BGM, their blood glucose levels, all from their smartphone. The promise of completing this “closed-loop” or “semi-closed-loop” system is clear, being able to pull all the information and data from the various key devices into a single platform for analysis, support and real-time decision making.
“As an early adopter of this new era of medical technology, Biocorp is in a prime position to design, develop and commercialise connected devices as part of its “design to production” approach…”
The main cited drawback of the add-on approach is that it adds an extra use-step for the patient. For example, the patient needs to remember to transfer Easylog from their used pen injector to the new one every time they change device. However, Biocorp has spent a lot of time ensuring that the process is as seamless as possible, making it really easy to put the add-on on the new device and requiring no further steps to prepare or activate it. The patient simply puts Easylog onto their device and then uses the pen injector as they usually would.
Easylog will be launched in 2019. At launch, it will be compatible with all major insulin pen platforms currently on the market and connect to any diabetes support platforms, thus fulfilling its purpose of facilitating treatment management for patients. Whilst Easylog will initially be targeted towards the diabetes market, it will in turn be adapted to other indications and markets. Biocorp has already begun tailoring of the technology to other therapeutic areas.
Service Provider Business Model
Rather than offering the Easylog add-on alone, Biocorp offers it as part of a fully integrated platform comprising a connected device, an app, and coaching services (Figure 3). This business model provides additional services to the patient, and real-time data that is valuable for payers (e.g. health plans and large companies in the US). Thus, the device is not an extra cost, but a central piece of a smart services offering. It is a model that is already proving profitable for some companies, for example Livongo (Mountain View, CA, US) and Omada (San Francisco, CA, US).

Figure 3: The service provider business case – integrated platform comprising device, app and coaching services.
EMBEDDED CONNECTIVITY TO IMPROVE EFFICACY
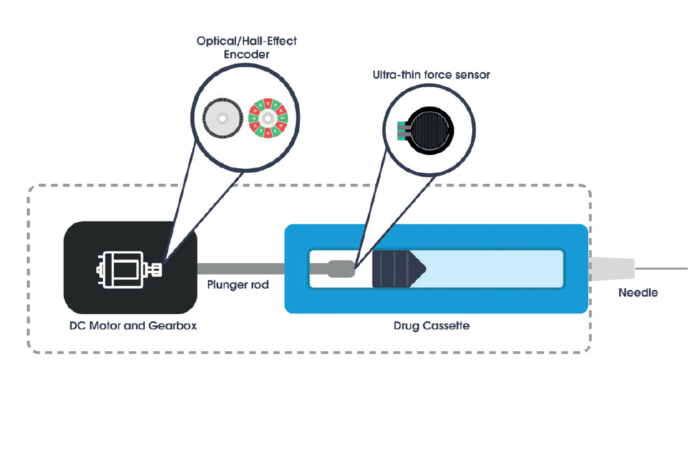
Biocorp also has products that take the integrated approach to connectivity, seamlessly building it in as a fundamental part of the design. They include the Datapen™ reusable pen injector (Figure 4) and the Onejet™ disposable autoinjector (Figure 5). The advantage of the integrated approach to connectivity is its ease-of-use and convenience for patients. By embedding the connected technology within the device, it can be made a more intuitive part of the user interface and this is the case with Datapen and Onejet.

Figure 4: Datapen, reusable pen injector.
Datapen is an electromechanical smart pen injector, compatible with standard cartridges and adaptable to dual-chamber cartridges as well. The electromechanical systems in Datapen provide a more comfortable, precise and easy injection for patients, and the digital elements manifest as an easily understood display with control buttons for optimal dialling and dosage, along with audio and visual cues to support the patient through the injection process.
With embedded Bluetooth, Datapen allows for real-time data transfer to companion software for tracking and recording injection data.
Onejet is the first motor-driven, disposable and Bluetooth-connected autoinjector for use with standard prefilled syringes (PFS) from 1–2.25 mL. Onejet is supplied assembled and ready to use by the patient which, along with its integrated connectivity, makes Onejet an extremely convenient product from the patient perspective. Onejet, utilises an easy and innovative pairing system to connect to a smartphone app, which receives real-time data from Onejet and processes it to provide treatment history and reminders for patients.

Figure 5: Onejet, motorised disposable autoinjector.
Onejet is designed for use with biologic medicines, so had been engineered to be easily customisable for different product profiles with varieties of volumes and viscosities. The device includes an in-built passive safety system to prevent needlestick injuries and the needle is hidden at all times, before, during and after injection, with needle insertion automatically triggered by a skin sensor. Like Datapen, Onejet features audio and visual signals to help the patient keep fully abreast of the progress of their injection.
TRAINING PATIENTS TO BETTER USE THEIR INHALER
Whilst historically, Biocorp’s interest and experience has been in the parenteral sector, it has expanded its connected add-on offering into the respiratory area with Inspair™ (Figure 6). Pressurised metered dose inhalers (pMDIs) are often misused by patients, so Inspair was developed as a smart sensing add-on device for standard pMDIs. The add-on captures airflow data as patients use their inhaler, providing feedback on usage, as well as advice on improving their technique.

Figure 6: Inspair, add-on sensor for pMDIs.
Inspair is a two-part device: a cap and a sensor. This design means that Inspair can be universalised across pMDIs as only the plastic mouthpiece needs to be customised to fit a given pMDI, which significantly reduces time-to-market. The design also does not interfere with regular inhaler usage, making it patient-friendly and easy to use. Inspair can also be adapted for other types of respiratory device, thanks to its modular design.
The data captured by Inspair can be fed back to the patient in a number of valuable ways. Key on the subject of proper adherence to medication is that it can be used to help the patient improve their hand-breath co-ordination. Poor patient performance in this respect negatively impacts the dose of medication delivered. Inspair can also feed back the inhaled dose and measurements of inhalation depth and speed, which for certain indications can be hugely valuable for tracking the course of treatment and enhancing ongoing decision-making with respect to patient care.
Much like Biocorp’s parenteral offerings, Inspair connects to a companion smartphone app via Bluetooth. The device captures and logs each actuation of the device, together with the time and date to provide patients and healthcare providers with an accurate treatment history, interwoven with assessment of quality of delivery. These features make Inspair a valuable addition to an inhalable offering, either as onboarding for new patients, a data capture tool for clinical trials or as part of a commercial product to help patients manage and track a chronic condition.
CONCLUSION
Connectivity is very much part of the future of drug delivery and healthcare as a whole, and with it will come new business models, opportunities and challenges. As an early adopter of this new era of medical technology, Biocorp is in a prime position to design, develop and commercialise connected devices as part of its “design to production” approach. With devices following both the add-on and integrated approaches to connectivity, Biocorp is ready to fulfil the opportunities presented by the new digital era.
REFERENCE
- Bazzoli F, “Top 10 predictions for healthcare IT in 2018”. Health Data Management, Dec 2017.