Citation: Popescu C, “Orally Disintegrating Films for the Delivery of Biotherapeutics & Nano/Microparticle Formulations”. ONdrugDelivery Magazine, Issue 77 (Jul 2017), pp 40-43.
Carmen Popescu summarises the benefits and applications of orally dissolving film dosage forms, with claims supported by two case studies, one demonstrating the suitability of ODFs for the delivery of micro and nanoparticles, the other showing how this property of ODFs exhibits promise as a next-generation immunisation system.
“This platform provides a non-invasive alternative to IV administration while producing excellent dose content uniformity and rapid dissolution performance. The ability to avoid first-pass metabolism makes ODF a very attractive delivery system, especially for paediatric and geriatric populations…”
Amongst patient-centric drug delivery systems designed to increase compliance in specific patient populations (paediatric, geriatric and psychiatric patients, and those with dysphagia, for example) orally dissolving films (ODFs) are preferred to classic dosage forms. Principally, this is due to their ease of administration without water (allowing portability, they can be taken “on the go”) and their pleasant taste and mouth feel – making them as much a “treat” as a treatment.
The main pharmaceutical appeal of ODFs as a class of drug delivery system resides in the fact that they can deliver the drug directly to the systemic circulation (avoiding first-pass metabolism). ODFs inherently provide for lower doses, thereby enhancing drug efficacy, improving the onset of action, and consequently patient compliance. Manufacturer’s attraction for these dosage forms resides in improved lifecycle management, market differentiation, innovation and brand creation.
In recent years, pharmaceutical companies are focusing their attention on continuous processing and ODFs are very good candidates when processed by hot melt extrusion (HME).
ODF FORMULATION REQUIREMENTS
Usually the film strip size is 2×3 cm at 30-40 mg weight and loaded with and 20-30 mg active pharmaceutical ingredient (API). The composition requires:
- A film forming polymer or combinations of polymers (e.g. starches, maltodextrins, pullulan, gelatine, cellulosic derivatives (HPMC), alginate, carrageenan, gums, etc)
- A plasticiser (e.g. sorbitol, glycerol, triacetin or propylene glycol)
- A sweetening agent
- A flavouring agent
- A colouring agent
- A surfactant, depending on formulation particulars
- A saliva stimulating agent.
BEST API CANDIDATES FOR ODF FORMULATION
Hydrosoluble and ethanol-soluble APIs are ideal candidates at a low dose of 5-30% w/w of the dry substance in the formula, meaning less than 30 mg/day dosing. Highly potent drugs and those prone to first-pass metabolism can be delivered at low doses in ODFs. Recently (as in the case studies that follow) nano/micronised BCS class II and IV drugs and biomolecules have also been identified as suitable candidates for ODF delivery, replacing an injectable form with an oral one by selecting film forming polymers with suitable rheological properties.
WHAT PROCESSES ARE AVAILABLE FOR MANUFACTURING ODFS?
Commonly used methods in the industry are:
- Solvent casting
- Hot melt extrusion (HME)1
- Solid dispersion
- Semisolid casting
- Rolling.
In recent years 2D and 3D printing methods are making rapid progress by facilitating the translation of ODFs from patient-centric to truly personalised medicine.
A major challenge in ODF formulation is excipients screening which must be done in order to find the right balance between disintegration time, rheological properties, API stability and mouth feel (see case studies that follow) while minimising the number of ingredients. It is better to handle just one polymer rather than a combination of polymers.
CASE STUDY 1: ODF FOR BENZOCAINE MICRO / NANOPARTICLE DELIVERY
Micronisation and nanonisation are methods used to increase the aqueous solubility of BCS class II and IV drugs. Traditionally, nanoparticles are delivered by the intravenous route, meaning patient compliance is impaired. We found that ODFs are a very good oral delivery alternative for nano and microparticles due to the convenience of administration, dose uniformity and physical stability.
Using Roquette’s single, non-GMO pre-gelatinised hydroxypropyl pea starch polymer, Lycoat® RS720, ODFs containing benzocaine as a model drug were prepared using a film casting technique at room temperature. Lycoat® RS720 dispersed easily in cold water within minutes without lump formation. It is able to form films without the need for organic solvents, and permits loading of API (benzocaine as a model drug) in crystalline form as micro/ nanoparticles. Lycoat® RS720 has a neutral taste and colour.
Drug Content Uniformity
ODF strips were completely dissolved in 100 mL HCl pH 1.5 under sonication and then filtered through a 0.2 μm syringe filter. The drug concentration was evaluated by UV absorption at 226 nm, in triplicate, using a UV-Vis spectrometer Lambda 20 (Perkin Elmer, Waltham, MA, US). Both micro and nanoparticle strips exhibited good content uniformity as evidenced in the concentration standard deviation (Table 1).
| Sample ID | Strip mass | Benozocaine mass/strip | Benozocaine concentration |
| mg (SD) | mg (SD) | w/w% (SD) | |
| Micro | 80.8 (10.7) | 10.4 (1.9) | 12.9 (0.5) |
| Nano | 53.2 (1.4) | 4.0 (0.1) | 7.5 (0.2) |
Table 1: Benzocaine ODF drug content uniformity.
Dissolution Evaluation
Simulated saliva consisted of a phosphate buffered saline solution (2.38 g Na2HPO4, 0.19 g KH2PO4 and 8 g NaCl per litre of distilled water adjusted with phosphoric acid to pH 6.75). Dissolution profiles of benzocaine ODFs were obtained using a DISTEK (Rainbow Dynamic Dissolution Monitor System coupled with Indigo data process software) in 500 mL of simulated saliva fluid at 37 ±0.5 °C with stirring at 100 rpm. Benzocaine ODFs were coupled with a pin and the dissolution process was progressed at the bottom of a vessel. The drug concentration was evaluated by UV absorbance at 282nm, in triplicate, using a UV-Vis spectrometer Lambda 20, Perkin Elmer (Figure 1).

Figure 1: Benzocaine ODF Dissolution.
Rheological Properties Evaluation
Young’s modulus, tensile strength and elongation at break were determined using an INSTRON 4502 universal testing machine (Instron, Norwood, MA, US), equipped with two pneumatic grips. The ODFs were placed between the grips and tensile stress was applied at 50 mm/min until rupture (Table 2).
| Thickness (mm) | Tensile strength (MPa) | Elongation at break (%) | Young’s modulus (MPa) | |
| ODF placebo | 0.098 | 12.8 | 2 | 575 ± 35 |
| Benzocaine ODF | 0.102 | 6.5 | 2 | 195 ± 60 |
Table 2: Benzocaine ODF rheological properties.
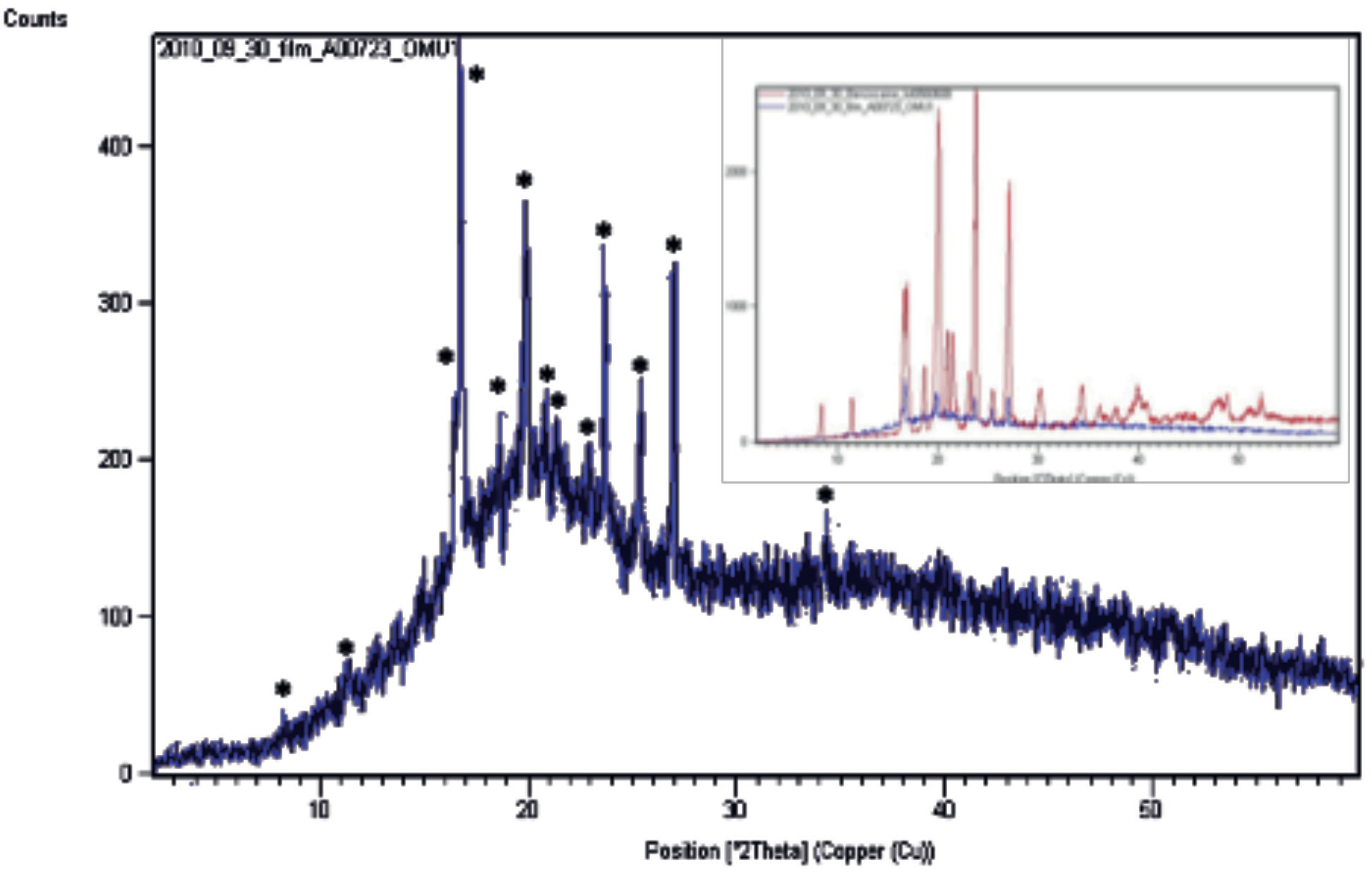
Powder X-ray diffraction (PXRD)
Data were collected using an X’Pert Pro MPD system (PANalytical, Almelo, the Netherlands) equipped with a copper anode (Kɑ = 1.5406 Å), programmable divergence slit and X’Celerator™ RTMS detector. The operational voltage and amperage were set to 45 kV and 40 mA, respectively.
Diffraction data were collected over a 2-60° 2Ө at a step size of 0.0170° and an irradiation time of 31.75 seconds/step. The benzocaine powder sample was back-filled into a stainless steel spinning sample holder, while the benzocaine ODF was placed into a zerobackground stainless steel sample holder. PXRD data for Benzocaine ODF confirm the presence of crystalline API (Figure 2). Significant diffuse scattering over 2Ө range is representative of polymer film matrix.
The Figure 2 inset shows the superimposition of benzocaine PXRD pattern (red) with film pattern (blue) indicating benzocaine diffraction peaks in the film pattern.

Figure 2: Benzocaine ODF powder X-ray diffraction.
Summary
ODF technology is a classical dosage form which has been shown to be a novel vector for delivering micro and nanoparticles of BCS II and IV drugs. This platform provides a non-invasive alternative to IV administration while producing excellent dose content uniformity and rapid dissolution performance. The ability to avoid first-pass metabolism makes ODF a very attractive delivery system, especially for paediatric and geriatric populations.
CASE STUDY 2: ODF BUCCAL MEASLES VACCINE FOR NEXTGENERATION IMMUNISATION
The buccal cavity possesses a rich source of antigen presenting cells (APCs) such as dendritic cells, which can be harnessed to immunise against infection. Immunisations using ODFs could be a very viable route in paediatrics.2
Live attenuated measles virus (antigen) and alum (adjuvant) were added to the solution (BSA crosslinking with glutaraldehyde) and spray dried using the Büchi Spray Dryer B-290 (Büchi, Flawil, Switzerland). Resulting microparticles (MP) were then incorporated in the ODF formulation based on Lycoat® RS720 film forming polymer (in presence of plasticiser) at room temperature under continuous mixing for 10-15 minutes until the suspension was uniformly dispersed.
The yield of the vaccine nanoparticles was 84.6% w/w with the size range (Malvern particle size analyser, Malvern Instruments, Malvern, UK) of 0.5-0.9 μm with a mean size of 0.67 μm. The half time of release, i.e. the time taken to release 50% of vaccine antigen from the nanoparticle, was seen to be about 12 hours. The rheological values of the vaccine loaded in oral disintegrating film (ODF) are summarised in Table 3.
| Tensile strength (MPa) | Young’s modulus (MPa) | Elongation (%) | |
| Average | 158 | 739 | 122 |
| Deviation | 24 | 192 | 4 |
Table 3: Measles vaccine nanoparticles ODF rheological properties.
In Vivo Immunisation Results
The efficacy of the measles vaccine ODF was tested in vivo in two pigs by delivery via the buccal route. Blood serum samples were collected every two weeks and a specific ELISA was performed to quantify the amount of specific antibody present. The antibody titres from the in vivo immunisation studies are shown in Figure 3. There was a significant increase in the antibody level after weeks 2, 4 and 6, compared with pre-dose levels.
APCs and Co-Stimulatory Molecule Expression on Dendritic Cells

Figure 3: Serum antibody levels (n= 4). The serum antibody levels were measured using ELISA. There was a significant increase in the IgG level post dosing when compared with pre-dosing (P < 0.01).
The antigen presentation is seen on either MHC I and II molecules. Along with this, there is a co-stimulation which is required for the APC to induce a Th1 or Th2 response. The dendritic cells were exposed to the vaccine microparticles for 24 hours and MHC I and II along with CD80 and CD40 (co-stimulatory molecules) were measured using flow cytometry (Figure 4). There was a significant increase in antigen presentation and co-stimulatory molecule expression on the APCs.

Figure 4: Antigen expression and co-stimulatory molecule expression on dendritic cells after exposure to the vaccine microparticles and blank microparticles (24 hrs.).
Summary
Buccal delivery of ODFs loaded with vaccine nanoparticles is a promising immunisation system. Vaccine nanoparticles are better taken up by the antigen presenting cells leading to further downstream process, generation of antibodies and thus creating an effective immunisation strategy. These encouraging preliminary results, will lead the way for further research in this area.
GIVING PRACTICALITY TO NOVELTY
It is well known that more than 45% of new drug entities have solubility issues and micronisation / nanonisation is one way to address this problem. However, micro- and nano-scaled drugs are usually delivered as injectables. Why an ODF? As shown in the above two case studies a good film former polymer can accommodate both hydrosoluble and insoluble (nano / microparticle) APIs of small and large molecules in an accurate dose, without changing their physical morphology (size, charge, crystallinity, etc). As the next generation of drugs belong to biopharmaceuticals, ODFs can be the best option for their oral delivery in order to increase patient compliance.
REFERENCES
- Pimparade MB , Vo A, Maurya AS, Bae J, Morrott J, Feng X, Kim D W, Kulkarni V, Tiwari R, Vanaja K, Murthy R, Shivankumar HN, Neupane D, Mishra SR, Murty SN, Repka MA, “Development and evaluation of an oral fast disintegrating anti-allergic film using hot-melt extrusion technology”. Eur J Pharmaceutics Biopharmaceutics, 2017, Vol 119, pp 81-90.
- Gala RP, Popescu C, Knipp GT, McCain RR, Ubale RV, Addo R, Bhowmik T, Kulczar CD, D’Souza MJ, “Physicochemical and Preclinical Evaluation of a Novel Buccal Measles Vaccine”. AAPS PharmSciTech, 2017, Vol 18(2), pp 283-292.