To Issue 185
Citation: Kaufmann N, Schori M, “Bio-Based Plastics in Drug Delivery: From Material Equivalence to Systemic Decarbonisation”, ONdrugDelivery, Issue 185 (Apr/May 2026), pp 46–51.
Nadine Kaufmann and Michael Schori analyse the feasibility of introducing sustainable materials within current regulatory frameworks, demonstrating this with a range of Ypsomed products and illustrating ways to integrate sustainable principles further into the value chain.
CARBON FOOTPRINTS: AN INDUSTRY TREADS CAREFULLY
The pharmaceutical industry is facing growing pressure to reduce its carbon footprint, particularly regarding Scope 3 emissions and product lifecycle impact. Combination products, including self-injection devices used in chronic therapies, are increasingly visible in this discussion, as frequent administration over many years translates into substantial material consumption and end-of-life waste. Healthcare systems, investors and procurement bodies are beginning to scrutinise not only therapeutic outcomes but also environmental performance.
At the same time, drug delivery systems operate within one of the most tightly regulated engineering environments in the industry. Materials are qualified, validated and embedded within controlled specifications, often across global markets. Any modification must be supported by biocompatibility assessments and managed under rigorous change control.
“SUSTAINABILITY INITIATIVES MUST ACHIEVE ENVIRONMENTAL GAINS WITHOUT INTRODUCING TECHNICAL OR REGULATORY UNCERTAINTY.”
This creates a structural tension: decarbonisation calls for material evolution, while regulation prioritises stability and predictability. In this context, sustainability initiatives must achieve environmental gains without introducing technical or regulatory uncertainty.
Material Evidence: The Case for Regulation
Regulatory conservatism should not be understood as reluctance to innovate, but as an integral framework designed to safeguard patient safety and product reliability. Within this environment, a material is never just a material – it forms part of a validated system that interacts with a medicinal product, interfaces with the patient and is documented across technical files and regulatory submissions. Any proposed substitution, even when driven by sustainability objectives, must therefore be assessed through the same disciplined processes applied to safety and performance changes.
Combination products are particularly sensitive to material modifications. Polymer components may influence extractables and leachables profiles, sterilisation behaviour, mechanical stability and long-term ageing characteristics. Material choices are closely linked to ISO 10993 biocompatibility evaluations. Thus sustainability initiatives must operate within this governance structure. Procurement ambitions alone are insufficient; engineering, regulatory and quality functions must align on equivalence criteria, documentation expectations and lifecycle implications.
The central question is not whether a material is renewable, but whether it can be integrated without altering the validated safety and performance envelope of the device. In the last few years, several established self-injection platforms have answered this affirmatively, demonstrating that sustainability can be implemented within existing regulatory frameworks.
In practice, this approach is embedded within Ypsomed’s NetZero Program, which forms part of the company’s science-based emissions reduction roadmap targeting net zero across the value chain. Within this framework, selected flagship platforms, including YpsoMate 1.0, YpsoMate 2.25 and UnoPen, are available in carbon-reduced variants. These integrate bio-based plastics with International Sustainability and Carbon Certification (ISCC+) while maintaining full biocompatibility, functionality and safety. Because the polymer chemistry remains unchanged, these implementations require no additional testing or requalification. For pharmaceutical partners, this demonstrates that decarbonisation can be incorporated into established platforms without altering validated device performance or regulatory status (Figure 1).

Figure 1: CO2 reduction in device material when switching to bio-based polymers for Ypsomed’s NetZero Program product platforms (cradle-to-gate, according to ISO 14067, which addresses biogenic CO2 uptake).
What’s the Matter? A Question of Carbon, not Chemistry
Early efforts to improve the environmental profile of medical plastics are often focused on biodegradable or novel bio-derived polymers. While well intentioned, these materials have frequently introduced new chemistries with different mechanical properties, altered extractables profiles and uncertain long-term ageing behaviour, and were not compatible with existing recycling streams. In regulated drug delivery systems, such changes inevitably translated into additional validation requirements and regulatory complexity. Sustainability therefore became associated with technical risk.
“RATHER THAN MODIFYING POLYMER CHEMISTRY, DROP-IN BIO-BASED MATERIALS RETAIN THE EXACT SAME MOLECULAR STRUCTURE AS THEIR FOSSIL-BASED COUNTERPARTS.”
The current generation of bio-based polymers follows a different path. Rather than modifying polymer chemistry, drop-in bio-based materials retain the exact same molecular structure as their fossil-based counterparts. The distinction lies solely in the origin of the carbon feedstock used during production, not the chemical architecture.
Conventional polymers and engineering plastics are typically derived from fossil-based petrochemical feedstocks such as naphtha or methanol. In bio-based variants, renewable feedstocks, such as waste-based oils or other second-generation biomass, are introduced upstream in the refining process, resulting in the same essential petrochemicals as in the fossil route. Through established polymerisation routes, these inputs yield polymers with an identical molecular structure, identical additive packages and identical material specifications. Thus the resulting resin is chemically identical to the fossil-based material (Figure 2).

Figure 2: Identical molecular structure for bio- and fossil-based polymers.
Noting to Declare: Documenting Equivalence
If polymer chemistry and material specifications remain unchanged, the regulatory implications of transitioning to bio-based feedstock are significantly reduced. From a technical standpoint, equivalence is established through specification matching, supplier declarations and continuity of documentation. The feedstock origin does not alter the polymer structure; therefore, toxicological profiles, extractables strategies and functional performance data remain within the validated range.
Documentation discipline, however, remains essential. Extractables and leachables rationales must demonstrate that existing data remain applicable, while supplier qualification and audit documentation, particularly under ISCC+ certification schemes, must ensure traceability of renewable content claims. For platform-based devices developed within mature quality systems, this level of assessment is a familiar exercise rather than an exceptional hurdle.
Performance Review: Meeting Expectations
Polymer performance is determined by molecular architecture, not by the origin of the carbon used to create it. With bio-based polymers, the polymer backbone, molecular weight distribution and additive formulation remain exactly the same, resulting in a material that exhibits the same mechanical strength, dimensional stability and resistance to environmental stress cracking as its fossil-based equivalent. When these parameters remain constant, functional behaviour remains constant.
For injection devices, this equivalence is essential. Structural components must retain their geometry under load, during sterilisation and throughout long-term ageing. Resistance to ethylene oxide or gamma sterilisation depends on the stability of the polymer chain rather than the feedstock source.
New platforms offer the opportunity to integrate sustainable materials from the very beginning of the design phase. However, it is equally important to continue using well-established engineering plastics with proven long-term behaviour and reliable technical performance. By transitioning these materials to bio-based feedstocks, their sustainability profile can be further improved without compromising functionality or safety. Since 2021, all of Ypsomed’s new platforms, such as YpsoFlow and YpsoDot, follow this process.
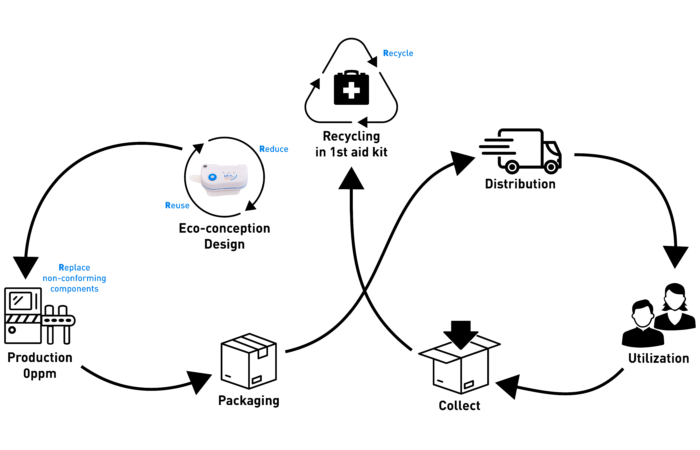
Yet, material strategy alone does not define the upper limit of sustainable device design. With YpsoLoop, an award-winning autoinjector combining sustainability and patient convenience, Ypsomed advances further. It combines carbon-reduced materials comprising only two bio-based, monomaterial subassemblies with a circular design philosophy, translated into a design made for automated disassembly and efficient recycling.
Putting Lifecycle in Scope
Material substitution is meaningful only when evaluated across the full product lifecycle. Product lifecycle assessments, conducted according to ISO 14040, 14044 and 14067 standards or Greenhouse Gas Protocol’s Corporate Standard, provide the framework for quantifying environmental impact from raw material extraction through manufacturing, distribution, use and end-of-life.
“A SHIFT IN FEEDSTOCK ORIGIN DIRECTLY REDUCES UPSTREAM CARBON INTENSITY AT ITS SOURCE, BUT REDUCTIONS MUST BE QUANTIFIED USING CLEARLY DEFINED CRADLE-TO-GATE SYSTEM BOUNDARIES TO ENSURE METHODOLOGICAL TRANSPARENCY.”
For device manufacturers, Scope 3 emissions typically dominate the carbon footprint, particularly those associated with raw material production. Design-stage decisions therefore exert disproportionate influence over total lifecycle impact. A shift in feedstock origin directly reduces upstream carbon intensity at its source, but reductions must be quantified using clearly defined cradle-to-gate system boundaries to ensure methodological transparency (Figure 3).

Figure 3: Carbon footprint of Ypsomed AG and its subsidiaries according to latest sustainability report.
Balancing the Books at Scale
The transition to renewable carbon does not occur at the moulding machine. It takes place upstream, at the refinery and feedstock level, where renewable raw materials are introduced into existing petrochemical infrastructure. Because physical segregation of molecules is neither practical nor necessary, attribution is managed through a mass balance methodology.
“YPSOMED PRIORITISES WASTE-BASED FEEDSTOCKS TO AVOID DEFORESTATION AND COMPETITION WITH FOOD CROPS.”
Under this approach, renewable feedstock input is tracked and allocated to specific outputted products. Independent certification bodies verify that defined volumes of renewable content enter the system and are correctly attributed downstream. ISCC+ certification provides chain-of-custody documentation, audit traceability and standardised accounting rules. ISCC+ also ensures a high standard of sources for bio-based materials protecting high-value land, ensuring eco-friendly production, upholding labour rights and complying with laws. Responsible sourcing is central to credibility. Ypsomed prioritises waste-based feedstocks to avoid deforestation and competition with food crops. They also exclude first-generation biomass routes to minimise land-use concerns (Figure 4).

Figure 4: The mass balance approach.
Ypsomed was first certified ISCC+ in 2023 and operates multiple certified sites globally. Net zero implementations within Ypsomed platforms rely on ISCC+ certified supply chains and standardised lifecycle assessments to substantiate carbon reduction claims. The environmental benefit lies in verified upstream carbon reduction, not in any physical difference in the moulded component.
Critical Mass: The GLP-1 Stress Test
The systemic relevance of bio-based integration becomes most visible in high-volume therapeutic areas. GLP-1 receptor agonists, widely used for type 2 diabetes and obesity, are frequently administered over extended periods, resulting in sustained high device usage across large patient populations. Millions of disposable injection devices are produced annually, each containing multiple polymer components derived from carbon-intensive feedstocks.
“DECARBONISING POLYMER INPUT REPRESENTS ONE OF THE FEW SCALABLE LEVERS AVAILABLE WITHOUT ALTERING CLINICAL PRACTICE.”
Lifecycle modelling of net zero variants demonstrates how per-unit reductions translate into substantial aggregate impact across global treatment volumes, reinforcing the value of integrating renewable carbon at platform scale. In such contexts, even incremental reductions in cradle-to-gate emissions per device compound rapidly at population scale. Extended dosing intervals may reduce injection frequency over time, but material intensity per device can remain similar. Therefore, decarbonising polymer input represents one of the few scalable levers available without altering clinical practice.
A Value Chain Reaction: The Need for Systemic Alignment
From a technical standpoint, drop-in bio-based polymers are no longer experimental. The chemistry is established, performance equivalence is demonstrable and regulatory pathways are well understood. The remaining constraints are largely systemic.
Renewable feedstocks compete with other sectors, including sustainable aviation fuels. Certification capacity must scale alongside demand. Regulatory recognition of mass balance attribution continues to evolve across jurisdictions. Pharmaceutical procurement frameworks may still prioritise short-term cost stability over lifecycle carbon metrics.
In this context, the limiting factor is not polymer science but market alignment. Decarbonisation requires a co-ordinated movement across resin suppliers, device manufacturers, pharmaceutical companies and healthcare systems. Technical feasibility alone does not guarantee adoption. Ypsomed engages with multiple stakeholders along the value chain, such as the Alliance to Zero of which it is a founding member, to collaboratively resolve outstanding questions and concerns.
Going Full Circle
The integration of bio-based polymers represents an important step, but it is only one facet of a deeper shift in how devices are conceived, designed and evaluated. As the industry matures in its understanding of lifecycle emissions and supply-chain decarbonisation, attention is increasingly moving towards design strategies that address impact at both ends of the product lifecycle.

Figure 5: YpsoFlow, YpsoDot and YpsoLoop: Since 2021, Ypsomed has applied ecodesign principles to all new product development.
At Ypsomed, responsibility is embedded as a core pillar of long-term strategy, shaping how devices are designed, manufactured and evaluated across their lifecycle. YpsoLoop is a tangible expression of this commitment, a platform conceived with circularity principles embedded from the outset. Bio-based polymers are used as the standard, but rather than treating end-of-life as an afterthought, YpsoLoop’s architecture simplifies material recovery. This is achieved through design for automated disassembly and efficient recycling, while retaining the usability and safety expected of prefilled, two-step autoinjectors. This approach acknowledges that reducing embodied carbon upstream is only part of the equation; true structural decarbonisation requires devices that are designed to feed back into material cycles once they have been used.
Future innovation in sustainable drug delivery will likely build on this template: combining low-impact feedstocks with device designs that anticipate and enable responsible end-of-life pathways. Collaboration across manufacturers, healthcare systems and recycling infrastructure providers will be essential for this next horizon of impact (Figure 5).
YpsoMate, UnoPen, YpsoFlow, YpsoDot and YpsoLoop are registered trademarks of Ypsomed AG. The product images shown are for illustrative purposes only. They are the property of Ypsomed AG and are used with permission. © 2026 Ypsomed. All rights reserved.