Citation: Sørensen B, “Advantages of Using an Electronic Injector for Dual Chamber Cartridges”. ONdrugDelivery Magazine, Issue 67 (May 2016), pp 14-18.
Bjarne Sørensen explores the benefits offered by an alternative technology – the electronic injector – and how it can be used to incorporate likely future requirements such as connectivity and reduced packaging.
Dual chamber cartridges (DCC), with separate chambers for the lyophilised product and the diluent, are becoming increasingly popular within the pharma industry and, compared with the traditional vial and syringe solution, potentially offer ease-of-use advantages for end users.
“An electronic injector offers significant enhancements to the user-friendliness of the whole drug administration process…”
Reconstitution typically happens by pushing the plunger unidirectionally into the syringe, so that the diluent transfers to the lyophilised product via a bypass in the glass, and the drug reconstitutes ready for injection. Most DCC cartridges come without any separate device but, in some cases, a specific manual device handles the reconstitution process separately from the injection device.
CHALLENGES OF DUAL CHAMBER CARTRIDGES
The preparation and injection process for DCCs can, under some circumstances, be a challenge for the end user:
- DCCs for high-volume applications tend to be quite long, given the sequential dual chambers, and can thus be a challenge to handle for patients with impaired dexterity, due to the overall length of the syringe and plunger.
- The viscosity of the drug can impose challenges because of the force required to push the plunger once the drug has been reconstituted.
- Limited solubility can impose certain time delays and mixing requirements before injection.
- The repeatability of reconstitution process is an issue – especially where the drug has a tendancy to foam, clump or where it may be damaged by over-enthusiastic agitation.
- Controlled orientation is required during the reconstitution – the needle must be pointing upwards.
STRATEGY FOR DEVELOPMENT
Medicom Innovation Partner has an extensive background within advanced electronic injection systems and connected services, including new technologies and concepts for electronic injectors for DDCs. These innovative technologies are being embodied in products for Medicom’s pharmaceutical customers.
It is important to consider the administration of the drug and the primary packaging very early in the drug development process. Medicom works with its customers on the long-term strategy for the drug and its delivery system, carrying out a 360-degree overview of the specific therapy to determine the optimum device strategy.
Ideally early enough in the lifecycle to influence and determine the optimum primary packaging, or with the already chosen primary packaging as a boundary condition. Part of our strategic review includes the consideration of electronic injectors for DCCs.
An electronic injector offers significant enhancements to the user-friendliness of the whole drug administration process.
THE KEY COMPONENTS OF ELECTRONIC INJECTORS
The technologies typically used in the electronic injector are all tried, tested and proven elements, and configurable to the exact requirements of the therapy and each case.
Motor-Driven Plunger
- The force needed by the patient is significantly reduced as operating the plunger simply requires the push of a button
- The device tightly controls speed of the plunger during reconstitution and injection. The speed can either be pre-determined by the device, or the user can be given control so that they can minimise any pain or discomfort
- The device can be set to deliver a full dose, or to deliver specific dose sizes as required
- Accommodation of large volume injections is straightforward
- A higher driving force is provided than can be reasonably expected from a patient so that administration of viscous drugs through narrow-gauge needles is possible
- The softly controlled start and stop of the plunger removes the typically sudden, jerky, noisy and uncomfortable release of forces from traditional mechanical injectors.
Electronic Architecture
- There is seamless opportunity for advanced control options, connectivity options and interfaces
- Orientation sensing can be built in the device, enabling control over the reconstitution and priming process, for example and, if required, the injection process
- Additional time to ensure solubility can be implemented
- A specific dwell time after injection can be implemented
- Various options for reminders to take the dose are possible
- Connectivity options are available such as Low Power Bluetooth to smart phones
- Radio-frequency identification (RFID) tags or 2D barcodes can be read to avoid counterfeit and read exact drug data
- The lifetime of an electronic injector can be set – e.g. to three years – and then be recycled in a controlled process.
Main building blocks
The usual building blocks of an electronic injector include (Figure 1):

Figure 1: Key components of the Electronic Injector – 1.4 mL DCC cartridge for size reference.
- A motor and spindle mechanism, which can drive the plunger forwards and backwards if necessary; the motor can be integrated with a small planetary gear
- A battery pack, which can enable a month of use between charging
- A control circuit with a small micro-processor that controls the overall functionality of the device and other peripheral elements like the connectivity interface
- A small display, if required by the functionality/therapy
- Buttons for controlling the device
- A motorised needle insertion, if required
- A hidden needle, with needle safety, if required Skin contact for automatic release of the injection
- A charging circuit – wired or wireless charging available, as required
- Provision to hold and exchange the cartridge, so that it can be changed effortlessly
- Provision to mount the needle, e.g. needles from the comprehensive diabetes portfolio.
FUTURE BENEFITS OF ELECTRONIC INJECTORS OVER OTHER TYPES
The flexible conceptual and technological opportunities of the electronic autoinjector provide significant opportunities for optimisations compared with traditional mechanical systems (Figure 2).

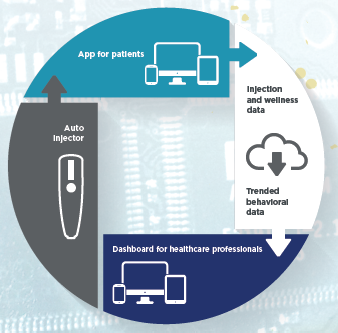
Figure 2: The complete connected system architecture.
“For example, a bi-weekly injection over three years, with a disposable autoinjector, leads to 312 complete sets of plastic components, springs, packaging etc thrown away, versus only one electronic injection device – and this is just for one patient…”
The built-in infrastructure and opportunities for connectivity, add a very interesting and future-orientated functionality to the electronic injector. By adding a Low Power Bluetooth interface in the injector, it is possible to communicate safely with a smartphone and, through an app developed and customised for the specific therapy, it is also possible to communicate with cloud storage and advanced data analytics. Such a system allows healthcare professionals, caregivers or other relevant stakeholders to access valid data of the drug usage, possibly also paired with behavioural data from the patients.
The electronic injector can uniquely convey exact and valid data about the drug administration, such as time of the injection, dose strength and volume, lot number etc, and it is now possible to create a coherent and consistent system approach to the drug delivery challenge, meeting the demands for game-changing innovation in the marketplace.
This is exactly the kind of functionality that will be hugely relevant, when the disruptive paradigm changes regarding demands for safe migration of treatment to home setting, outcome-driven drug pricing and demands for integration of drug delivery data with the health ecosystem, enter the marketplace in full force.
Another aspect of the electronic injector is that of waste management, which is of increasing importance throughout the industry and society. A reusable electronic injector can significantly limit the waste generated by the administration of the therapy, as the only discarded part is the primary packaging.
For example, a bi-weekly injection over three years, with a disposable autoinjector, leads to 312 complete sets of plastic components, springs, packaging etc thrown away, versus only one electronic injection device – and this is just for one patient.
The necessary production volumes for an electronic injector are thus also significantly smaller compared with a disposable autoinjector, where expensive multi-cavity tooling and automated assembly lines are often required due to the high production volumes. The production set-up for an electronic injector is typically single cavity tooling and universal assembly methods and equipment, which result in a much lower investment for tooling and equipment.
The cost structure of the drug is positively influenced by reducing the costs of parts that will be thrown away for each injection, increasing the competitive edge of the drug and affecting the reimbursement aspects positively.
CONCLUSION
Generally, it is widely recognised that user-centric devices and services:
- Improve efficacy and safety
- Reduce administration errors
- Improve patient & physician engagement and increases adherence
- Create brand loyalty, which leads to better patient and clinical retention
- Facilitate safe migration of treatment to home setting.
Medicom’s view is that disruptive technologies change the pharmaceutical industry for the better, but it does require a mind-set that does not conform to pre-existing standards and norms, and they are not necessarily able to be valued by traditional quantitative market research.
In this article we have focused on the electronic injector for DCCs, but many of the unique advantages will also be beneficial for other primary packaging types, for which Medicom is also developing or manufacturing similar electronic injector solutions and complete connected systems.