To Issue 184
Citation: Owen M, “Intranasal Drug Delivery: Formulation, Device Design and Characterisation Strategies”. ONdrugDelivery, Issue 184 (Apr 2026), pp 58–60.
Dr Matthew Owen reviews the formulation and device challenges, designs and considerations that are shaping the nasal delivery market.
Intranasal drug delivery has evolved into a clinically established route for both local and systemic therapies, with growing interest in targeted nose-to-brain applications.1 Once primarily associated with topical decongestants and corticosteroids, the nasal route is now used for rapid systemic delivery, rescue medications, peptides and central nervous system (CNS) therapeutics.2–5 This progression reflects advances in formulation science, device engineering and an improved understanding of nasal physiology.

Figure 1: Anatomical descriptions for nasal cavity delivery.
“THE NASAL CAVITY PROVIDES SEVERAL INHERENT ADVANTAGES FOR DRUG DELIVERY: RAPID ABSORPTION DUE TO HIGH VASCULARISATION, AVOIDANCE OF FIRST-PASS HEPATIC METABOLISM AND NON-INVASIVE ADMINISTRATION THAT SUPPORTS PATIENT ADHERENCE.”
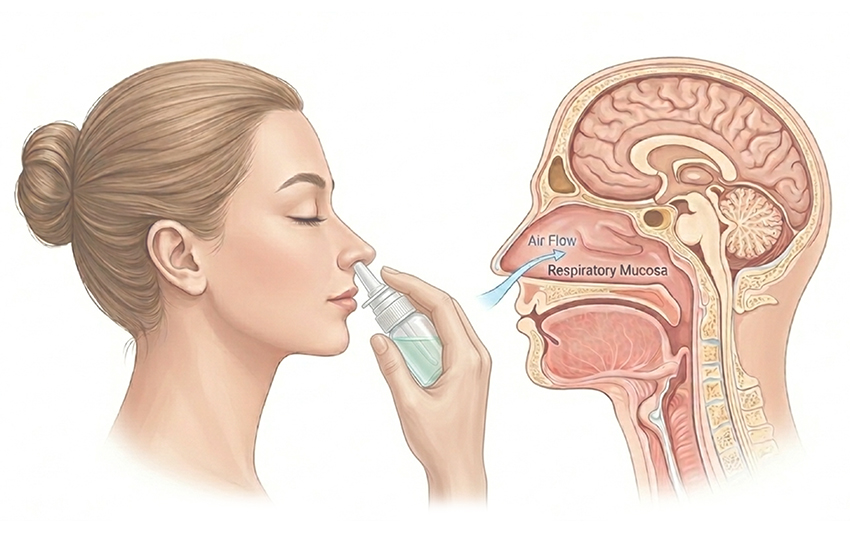
The nasal cavity can be broadly divided into vestibular, respiratory and olfactory regions (Figure 1). The vestibule primarily serves as an entry zone, with minimal contribution to absorption of drug product. The respiratory region, which includes the inferior, middle and superior turbinates, accounts for most of the functional surface area and is the principal site of systemic absorption. The nasal cavity provides several inherent advantages for drug delivery: rapid absorption due to high vascularisation, avoidance of first-pass hepatic metabolism and non-invasive administration that supports patient adherence.6,7 In addition, anatomical connections to the brain via the olfactory pathway and a network of nerves, known as the trigeminal pathway (Figure 2), present an opportunity for CNS targeting that can bypass the blood–brain barrier.1,2,8 Direct transport via the olfactory and trigeminal pathways is believed to contribute to CNS drug exposure, whereas drugs reaching the brain via systemic circulation must cross the blood–brain barrier. A drug that is swallowed may undergo gastrointestinal absorption and first-pass metabolism, minimising effective nasal bioavailability.

Figure 2: Targeted intranasal delivery pathways.
Despite these advantages, intranasal product development presents distinct technical challenges. The nasal cavity is a dynamic and protective environment characterised by limited dosing volume (approximately 100–150 μL per nostril), rapid mucociliary clearance, enzymatic activity and significant inter- and intra-patient variability.9 Therefore, effective development requires co-ordinated optimisation of formulation properties, regional deposition and device performance.
FORMULATION CONSIDERATIONS
Aqueous Liquid Nasal Sprays
Systemic delivery of therapeutics via the intranasal route is highly dependent on the physical and chemical properties of the formulation, nasal physiology and geometry. The key hurdles lie in the workable surface area, the rheological properties of the mucus layer, presence of enzymes and ciliary clearance.8–10 For systemic delivery, most of the formulation will deposit on the respiratory mucosa, which has an active surface area of approximately 150–180 cm², depending on the measurement method.11
The viscosity of the mucus layer can influence diffusion and residence time. Healthy mucus exhibits relatively low viscosity, but this can increase under diseased conditions, potentially affecting drug diffusion and clearance.12 Therefore, formulation flow behaviour and permeation into the mucus layer are important considerations, particularly with mucociliary transport.
Permeation enhancers can be added to the formulation to increase epithelial permeability and improve transmucosal transport. If extended residence time is desired, particularly for larger biomolecules, hydrophilic compounds, or CNS-targeted therapies, mucoadhesive compounds may be incorporated to prolong local retention.13–15
Other formulation factors include pH and tonicity modifiers. The local pH of the nasal mucosa is slightly acidic (5.5–6.5) in healthy humans and may become more alkaline (> 7) in diseased states, such as rhinitis.16 The formulation pH should be as close to physiological conditions as is feasible. Maintaining near-physiological pH supports ciliary function, minimises irritation and helps to preserve mucosal integrity.17
In terms of osmolality, the nasal mucosa is isotonic (approximately 290 mOsm/kg). Most nasal formulations are designed to approximate isotonic conditions to minimise changes in mucosal hydration, ciliary beat frequency and irritation.18,19 Hypertonic formulations may induce stinging, watering, sneezing or increased mucus secretion, potentially accelerating clearance.20
The addition of a preservative may be necessary to prevent microbiological contamination if the drug product is not produced in an aseptic environment or sterile filtered.21 Preservative selection depends on formulation pH and compatibility to ensure antimicrobial effectiveness and stability. As with all excipients, there are patient risks and side effects with each selection, especially with irritants and use with a targeted population such as children.17,21,22 Table 1 highlights critical key attributes and typical formulation additives.
| Attribute | Purpose | Typical Excipients |
| Permeation Enhancer | Improve permeability in viscous mucosa through typical paracellular or transcellular transport | • Alkyl saccharides (dodecyl maltoside, etc.) • Cyclodextrins (β-cyclodextrin, etc.) • Diethylene glycol monoethyl ether • Fatty acids (oleic acid, etc.) • Phospholipids (C16, C14, etc.) • Surfactants (PS20, PS80, etc.) |
| Mucoadhesive | Adhere to mucosa to minimise and prevent clearance | • Cellulose compounds (carboxymethyl cellulose, hydroxymethyl cellulose, etc.) • Polyethylene glycol polymers • Thiolated polymers • Poloxamers |
| pH Modifier | Target physiological pH to prevent irritation | • Buffers • Acids (HCl, etc.) • Bases (NaOH, etc.) |
| Osmolality Modifier | Target isotonic/low hypotonic environment | • Sugars (dextrose, sucrose, etc.) • Salts (NaCl, KCl, etc.) |
| Preservative | Prevent microbial growth and increase shelf life | • Benzalkonium chloride, chlorobutanol, benzyl alcohol, etc. |
Table 1: Critical key attributes of liquid nasal sprays.
Dry Powder Nasal Products
Dry powder-based nasal products represent an alternative approach with specific advantages and constraints. Powder formulations are often less compositionally complex than liquid sprays and, in some cases, may consist primarily of APIs with minimal excipients. Nasal powders are also advantageous for molecules with limited aqueous solubility or stability, such as biologics.23
A key difference from liquid systems is the engineering of the powders. The powders are commonly engineered using processes such as spray drying,24,25 which involve equipment configurations to ensure the desired particle size, morphology, dispersibility and inter/intra-active location. For nasal delivery, particle size distribution and aerodynamic behaviour are critical due to deposition and pulmonary safety considerations.13,14,25 For example, a powder with geometrical size distribution of 10–50 μm may still present a smaller aerodynamic diameter if the tap density is low, which increases the risk of unintended pulmonary deposition (<10 μm). Unintended pulmonary deposition may introduce additional safety considerations and should be minimised through particle engineering strategies.26
Typical nasal powder formulation may include APIs, permeation enhancers, salts and/or sugars, that are similar to excipients used in liquid systems.1,3,10 An additional key consideration is moisture control. Excipients should exhibit minimal hygroscopicity, so the manufacturing processes should minimise moisture exposure.25,27 Elevated moisture content can negatively affect powder dispersibility and reduce delivered dose performance.28 Dry powder systems often demonstrate improved chemical stability relative to aqueous formulations and may support an extended shelf life under appropriate storage conditions.29
Device Selection and Spray Performance
There are many forms and brands for unit-, bi- and multidose nasal device formats. Examples of US FDA-approved unit-dose products include Narcan (naloxone hydrochloride; Emergent BioSolutions, Gaithersburg, MD, US), RiVive (naloxone; Harm Reduction Therapeutics, Bethesda, MD, US), Zavzpret (zavegepant; Pfizer) and Neffy (adrenaline (epinephrine) – ARS Pharmaceuticals, San Diego, CA, US).
FDA-approved bi-dose products include niche products such as Spravato (esketamine – Janssen Pharmaceutical) and recently CARDAMYST (etripamil) was approved by the FDA for Milestone Pharmaceuticals (Quebec, Canada) using a bi-dose format. There are many multidose nasal over-the-counter medicines, such as Flonase (fluticasone; Haleon, Weybridge, UK) and Nasonex (mometasone furoate; Organon Pharma, Jersey City, NJ, US). For nasal powders, unit-dose devices are common, an example being BAQSIMI (glucagon; Amphastar Pharmaceuticals, Rancho Cucamonga, CA, US).
Dose strength selection and intended nasal target region are critical design considerations. Most devices are engineered to promote deposition within the respiratory mucosa to facilitate transmucosal systemic absorption (Figure 3).10,11,15,30,31

Figure 3: Plume characteristics for systemic and nose-to-brain delivery.
For systemic delivery, the respiratory epithelium, including the inferior, middle and superior turbinates, is the primary target. To promote broad coverage, plume width (W) should be relatively wider with a shorter plume length (L), generating a broader plume angle (φ). Spray velocity (v) should be sufficient to achieve deposition without excessive impaction.32,33 Droplet sizes are typically engineered to promote nasal deposition while minimising inhalation risk. Proper device orientation remains important to ensure consistent and reproducible deposition within the respiratory region, which comprises the majority of the active target area.
For nose-to-brain targeting, the olfactory regions represent less than 5% of the total surface area. Therefore, design controls in the device and dosing need to be established for maximum coverage.17,30 For instance, to increase the probability of olfactory deposition, the device and head placement should be tuned, such that the direction of the resulting spray is fine targeted to the olfactory region. A narrower plume angle and controlled velocity may reduce anterior impaction and improve olfactory targeting. Droplet size must be optimised to balance regional targeting with safety considerations.10,15,30
In Vitro Anatomical Models
An important tool for performance evaluation is the in vitro anatomical nasal cast model. These models are typically derived from single or average human anatomies using cyclic tomography datasets. They enable the quantification of regional deposition in areas of the nasal cavity, such as the vestibule, turbinates, olfactory and nasopharynx.34 Standard models are often constructed from rigid materials, such as polymers or stainless steel; however, a more representative nasal cavity would simulate a mucosal-like surface. This is typically achieved by using a viscous polymer or a combination of viscous polymer and surfactant.35 Such models significantly streamline product development and support formulation-device optimisation prior to clinical evaluation.
Characterisation and Performance Testing
The typical testing regimen includes both bulk formulation testing and device performance evaluation. These are usually performed in parallel or before formulation-device performance testing, such as assays of active and excipients (Table 2).
| Domain | Test | Liquid Nasal Spray | Nasal Powder | Clinical and Regulatory Relevance |
| Chemistry – Identification, Assay and Strength | Assay of active (HPLC/UPLC) | ✔ | ✔ | Label claim verification |
| Related substances and impurities (HPLC/UPLC) | ✔ | ✔ | Degradation and stability monitoring | |
| Assay of critical excipients (HPLC/UPLC/GC) |
✔ | ✔ | Label claim confirmation | |
| pH | ✔ | N/A | Nasal tolerability and stability assurance | |
| Osmolality | ✔ | N/A | Mucosal compatibility | |
| Moisture content | N/A | ✔ | Powder flow and stability control | |
| Blend uniformity | N/A | ✔ | Dose homogeneity assurance | |
| Physical Characterisation (Bulk) | Viscosity | ✔ | N/A | Flowability and spray performance |
| Particle size distribution (laser diffraction) |
N/A | ✔ | Spray performance and dispersion control |
|
| Density (bulk/tapped) |
✔ (Bulk) |
✔ (Tapped) |
Manufacturability and fill consistency | |
| Appearance | ✔ | ✔ | Visual quality control | |
| Foreign particulate matter (HiAC/microscopy) |
✔ | ✔ | Particulate safety assurance | |
| Residual solvent | ✔ | ✔ | Patient safety assurance | |
| Microbiological Control | Bioburden | ✔ | ✔ | Prefill microbial safety |
| Preservative content | ✔ | ✔ | Microbial integrity control | |
| Device Performance | Delivered dose | ✔ | ✔ | Dose accuracy |
| Pump delivery | ✔ | ✔ | Dose precision and reproducibility | |
| Dose content uniformity | ✔ | ✔ | Dose uniformity assurance | |
| DSD (Dv10, Dv50, Dv90) | ✔ | N/A | Deposition control and respirable fraction safety |
|
| Aerodynamic particle size distribution (APSD) | N/A | ✔ | Deposition control and respirable fraction safety |
|
| Spray pattern/plume geometry | ✔ | ✔ | Nasal cavity coverage | |
| Actuation force | ✔ | ✔ | Actuation consistency and patient usability |
|
| Nasal cast modelling | ✔ | ✔ | In vitro deposition characterisation | |
| Stability | Product stability | ✔ | ✔ | Shelf-life determination |
| Weight loss | ✔ | ✔ | Container-closure integrity | |
| Extractables and leachables | ✔ | ✔ | Toxicological risk mitigation |
Table 2: Integrated bulk and device characterisation strategy for nasal spray and nasal powder products.
A key difference between liquid and dry powder systems is the emitted particle size distribution. For liquid sprays, droplet size distribution is commonly measured via laser diffraction, which is the size distribution over a stable region in the timeframe of the spray. The key reporting parameters include Dv10, Dv50, Dv90, span and percent volume less than 10 μm.
These metrics characterise droplet distribution and support pulmonary safety assessments. This technique could be used for nasal powders, but the common practice is to use impaction techniques, such as next-generation impaction.36,37 Deposition across stages, including induction port or nasal inlet/globe, pre-separator, collection cups and final external filter, enables the calculation of the mass median aerodynamic diameter, fine particle fraction/dose and emitted dose. Using the size cut-offs from these stages enables the determination of the pulmonary safety risk of the nasal powder.
FUTURE OUTLOOK AND CONCLUSIONS
Intranasal drug delivery has become a practical and clinically established route for systemic and CNS-enabling treatments. As outlined, successful development requires careful alignment between formulation properties, particle or droplet engineering, device design and understanding of nasal anatomy. Small changes in viscosity, moisture content, plume geometry or particle size can meaningfully impact deposition and overall product performance. For both liquid sprays and dry powders, bulk chemistry and device function are tightly connected and must be developed together.
Looking forward, progress in this space will likely come from better integration of formulation and device development, along with more predictive in vitro tools, such as anatomically relevant nasal casts and computational modelling. Interest in peptides, biologics and other complex molecules will continue to push innovation, particularly for powder-based systems where stability metrics are advantageous. At the same time, variability in nasal physiology and the challenges of regional targeting, especially for CNS delivery, remain important areas for continued focus.
Overall, intranasal delivery is located at the intersection of formulation science, device engineering and nasal physiology. As these disciplines become more tightly co-ordinated, the nasal route is positioned to play an increasingly important role in rapid systemic therapies and future brain-targeted treatments.
REFERENCES
- Koo J, Lim C, Oh KT, “Recent advances in intranasal administration for brain-targeting delivery: a comprehensive review of lipid-based nanoparticles and stimuli-responsive gel formulations”. Int J Nanomed, 2024, Vol 19, pp 1767–1807.
- Qiu Y et al, “The nasal–brain drug delivery route: mechanisms and applications to central nervous system diseases”. MedComm, 2025, Vol 6(6), art 70213.
- Wei S et al, “The review of nasal drug delivery system: the strategies to enhance the efficiency of intranasal drug delivery by improving drug absorption”. Int J Pharm, 2025, Vol 676, art 125584.
- Peters JM et al, “Taking a newer, faster, intranasal route: a narrative review of transitioning to a less-invasive rescue treatment for seizure clusters”. Patient Prefer Adherence, 2024, Vol 18, pp 383–389.
- Alabsi W et al, “Nose-to-brain delivery of therapeutic peptides as nasal aerosols”. Pharmaceutics, 2022, Vol 14(9), p 1870.
- Tai J et al, “Different methods and formulations of drugs and vaccines for nasal administration”. Pharmaceutics, 2022, Vol 14(5), p 1073.
- Al-Hajaj N, Khalil R, Husseini GA, “Intranasal drug delivery: pathways, challenges, and advancements in CNS targeting”. J Drug Deliv Sci Technol, 2025, Vol 107, art 106825.
- Han SA et al, “The olfactory system: basic anatomy and physiology for general otorhinolaryngologists”. Clin Exp Otorhinolaryngol, 2023, Vol 16(4), pp 308–316.
- Chung S et al, “The nose has it: opportunities and challenges for intranasal drug administration for neurologic conditions including seizure clusters”. Epilepsy Behav Rep, 2023, Vol 21, art 100581.
- Gänger S, Schindowski K, “Tailoring formulations for intranasal nose-to-brain delivery: a review on architecture, physico-chemical characteristics and mucociliary clearance of the nasal olfactory mucosa”. Pharmaceutics, 2018, Vol 10(3), art 116.
- Gizurarson S, “Anatomical and histological factors affecting intranasal drug and vaccine delivery”. Curr Drug Deliv, 2012, Vol 9(6), pp 566–582.
- Masiuk T, Kadakia P, Wang Z, “Development of a physiologically relevant dripping analytical method using simulated nasal mucus for nasal spray formulation analysis”. J Pharm Anal, 2016, Vol 6(5), pp 283–291.
- Trenkel M, Scherließ R, “Optimising nasal powder drug delivery – characterisation of the effect of excipients on drug absorption”. Int J Pharm, 2023, Vol 633, art 122630.
- Vidgren MT, Kublik H, “Nasal delivery systems and their effect on deposition and absorption”. Adv Drug Deliv Rev, 1998, Vol 29(1–2), pp 157–177.
- Gao M, Shen X, Mao S, “Factors influencing drug deposition in the nasal cavity upon delivery via nasal sprays”. J Pharm Investig, 2020, Vol 50, pp 251–259.
- England RJ et al, “Nasal pH measurement: a reliable and repeatable parameter”. Clin Otolaryngol Allied Sci, 1999, Vol 24(1), pp 67–68.
- Nguyen LTT, Duong VA, “Nose-to-brain drug delivery”. Encyclopedia, 2025, Vol 5(3), art 91.
- Huijghebaert S, Hoste L, Vanham G, “Essentials in saline pharmacology for nasal or respiratory hygiene in times of COVID-19”. Eur J Clin Pharmacol, 2021, Vol 77(9), pp 1275–1293.
- Min YG et al, “Hypertonic saline decreases ciliary movement in human nasal epithelium in vitro”. Otolaryngol Head Neck Surg, 2001, Vol 124(3), pp 313–316.
- Shoseyov D et al, “Treatment with hypertonic saline versus normal saline nasal wash of pediatric chronic sinusitis”. J Allergy Clin Immunol, 1998, Vol 101(5), pp 602–605.
- Klöcker N et al, “Antimicrobial safety of a preservative-free nasal multiple-dose drug administration system”. Eur J Pharm Biopharm, 2004, Vol 57(3), pp 489–493.
- Kim BG et al, “Nasal pH in patients with chronic rhinosinusitis before and after endoscopic sinus surgery”. Am J Otolaryngol, 2012, Vol 34(5), pp 505–507.
- Cipolla D et al, “Inhalable and nasal biologics: analytical, formulation, development, and regulatory considerations”. J Aerosol Med Pulm Drug Deliv, 2025, Vol 38(5), pp 284–303.
- Su J et al, “Visualization of nasal powder distribution using biomimetic human nasal cavity model”. Acta Pharm Sin B, 2024, Vol 14(1), pp 392–404.
- Henriques P, Fortuna A, Doktorovová S, “Spray dried powders for nasal delivery: process and formulation considerations”. Eur J Pharm Biopharm, 2022, Vol 176, pp 1–20.
- Baltz N et al, “Advancing nasal formulation characterization: considerations toward a robust and precise method to determine the mass fraction below 10 μm in nasal products”. Aerosol Sci Technol, 2024, Vol 58(11), pp 1305–1317.
- Ehrick JD et al, “Considerations for the development of nasal dosage forms” in “Sterile Product Development” (Kolhe P, Shah M,Rathore N, eds). Springer, AAPS Series, 2012, Vol 6, pp 99–144.
- Jüptner A, Scherließ R, “Investigation of powder properties and application aspects impacting nasal deposition of spray-dried powders in a nasal cast”. Eur J Pharm Biopharm, 2025, Vol 209, art 114666.
- Fasiolo LT et al, “Opportunity and challenges of nasal powders: drug formulation and delivery”. Eur J Pharm Sci, 2018, Vol 113, pp 2–17.
- Djupesland PG, “Nasal drug delivery devices: characteristics and performance in a clinical perspective-a review”. Drug Deliv Transl Res, 2013, Vol 3(1), pp 42–62.
- Inthavong K et al, “A fluid and particle mechanics perspective of nasal drug delivery”. Expert Opin Drug Deliv, 2025, Vol 22(12), pp 1811–1828.
- Maaz A, Blagbrough IS, De Bank PA, “In vitro evaluation of nasal aerosol depositions: an insight for direct nose to brain drug delivery”. Pharmaceutics, 2021, Vol 13(7), p 1079.
- Foo MY et al, “The influence of spray properties on intranasal deposition”. J Aerosol Med, 2007, Vol 20(4), pp 495–508.
- Williams G, Suman JD, “In vitro anatomical models for nasal drug delivery”. Pharmaceutics, 2022, Vol 14(7), p 1353.
- Duong K et al, “Characterization of spray-dried powders using a coated Alberta idealized nasal inlet”. J Aerosol Med Pulm Drug Deliv, 2025, Vol 38(1), pp 1–12.
- Mitchell JP, “Particle size measurements from orally inhaled and nasal drug products”. J Aerosol Med Pulm Drug Deliv, 2021, Vol 34(6), pp 325–345.
- Copley M, Kippax P, “From actuation to deposition: particle sizing techniques for characterizing nasal drug delivery systems”. Inhalation Magazine, Apr 2012.