To Issue 184
Citation: Watts A, Wilcox D, “Expanding Horizons in Liquid Nasal Drug Development”, ONdrugDelivery, Issue 184 (Apr 2026), pp 22–28.
Dr Alan Watts and David Wilcox examine the current product designs in the nasal market landscape and outline key technical and regulatory considerations for liquid nasal drug products.
Over the past two decades, liquid nasal dosage forms have evolved from simple antihistamine or decongestant sprays into highly engineered drug-device combinations capable of addressing diverse therapeutic needs. Initially focused on local indications such as allergic rhinitis or intranasal influenza vaccines, the modality now supports products requiring rapid systemic absorption, caregiver-administered emergency therapies and, increasingly, direct nose-to-brain delivery.
This expansion reflects advances in formulation science – including solubility enhancement, mucoadhesive polymers and stabilisation of biologics – paired with sophisticated multidose and unit-dose device technologies. Regulatory frameworks have also evolved to recognise the clinical and public health value of nasal therapeutics, reinforcing the need for robust chemistry, manufacturing and controls (CMC) strategies.
Drawing on experience across development, clinical supply and commercial manufacturing, a co-ordinated approach to early CMC strategy enables streamlined scale-up, risk mitigation and regulatory readiness.
INDICATIONS AND EMERGING MARKETS
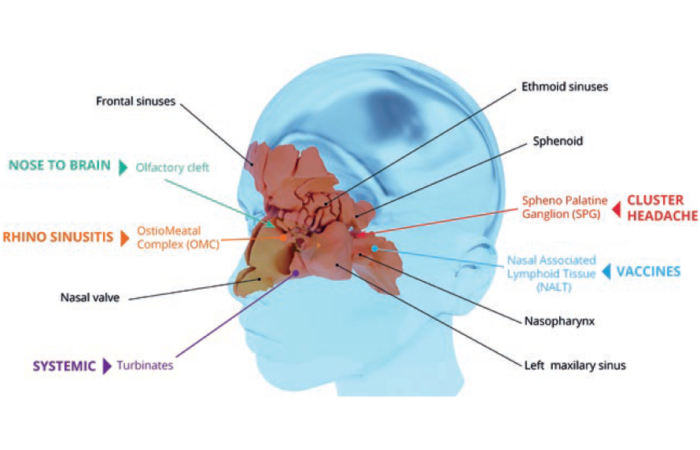
Liquid nasal delivery spans locally acting therapies, rapid systemic rescue medications and central nervous system (CNS)-targeted products. Established indications, such as allergic rhinitis and intranasal influenza vaccination, continue to represent substantial global volume. These products typically employ aqueous solution or suspension formulations delivered through high-precision multidose pumps. From a CMC standpoint, they require tight control of droplet size distribution (commonly Dv50 ~40–120 µm), spray pattern geometry, plume angle, priming/repriming behaviour and delivered dose uniformity across the container lifespan. Microbial control strategies – either preservative-based or preservative-free with sterile filtration and closed-tip systems – must support in-use stability. Extractables and leachables, container-closure integrity and ICH stability performance remain foundational development considerations.
“ACUTE PAIN MANAGEMENT PRODUCTS HIGHLIGHT THE NASAL ROUTE’S CAPACITY FOR RAPID SYSTEMIC ABSORPTION.”
Acute pain management products highlight the nasal route’s capacity for rapid systemic absorption. The highly vascularised respiratory epithelium (~150–200 cm² surface area) supports onset within minutes for appropriately designed small molecules. For moderately lipophilic agents, such as butorphanol or ketorolac, absorption occurs primarily via passive transcellular diffusion. Formulation strategies typically involve solubility enhancement (co-solvents or cyclodextrins), pH optimisation to increase the unionised fraction and viscosity modifiers to balance mucosal residence time against mucociliary clearance (approximately 15–30 minutes).
Similar principles underpin intranasal naloxone for opioid overdose rescue, where reliable pharmacokinetics under real-world conditions are essential. These emergency-use products have shaped regulatory expectations for unit-dose systems, emphasising delivered dose accuracy, shot weight reproducibility, spray content uniformity, human factors validation and device robustness.
Seizure rescue therapies have expanded outpatient management through caregiver-administered intranasal benzodiazepines. Because compounds such as midazolam and diazepam exhibit limited aqueous solubility, formulations frequently rely on non-aqueous systems, solubilising excipients and viscosity-modifying polymers while minimising irritation and run-off. Clinical data demonstrate achievement of therapeutic plasma concentrations within approximately 10–15 minutes.1
Other CNS-directed therapies further illustrate the platform’s evolution. Intranasal esketamine for treatment-resistant depression underscores the importance of deposition control and absorption kinetics. While the quantitative contribution of direct nose-to-brain transport remains debated, evidence supports the feasibility of olfactory and trigeminal pathways when formulation and device variables are optimised.2
“REPRODUCIBLE UPPER NASAL CAVITY DEPOSITION DEPENDS ON DROPLET SIZE, PLUME DYNAMICS AND ACTUATION CONSISTENCY, WITH SOME PROGRAMMES USING MECHANICALLY ASSISTED DELIVERY TECHNOLOGIES.”
Reproducible upper nasal cavity deposition depends on droplet size, plume dynamics and actuation consistency, with some programmes using mechanically assisted delivery technologies. Migraine therapies and emerging cardiovascular and emergency indications reflect continued innovation. Modern dihydroergotamine systems and calcitonin gene-related peptide receptors (CGRP)-targeted agents need predictable pharmacokinetics during acute episodes when oral absorption is compromised. Programmes in anaphylaxis and paroxysmal supraventricular tachycardia require ultrarapid systemic onset; for example, intranasal adrenaline (epinephrine) demands mitigation of oxidative degradation to achieve delivery performance comparable to autoinjectors.
Perhaps most transformative is the growing pipeline of peptides and proteins (Table 1). Large molecules face enzymatic degradation, limited epithelial permeability and rapid clearance. Strategies under investigation include mucoadhesive polymers, enzyme inhibitors, permeation enhancers and targeted deposition to the olfactory region. Intranasal insulin and glucagon-like peptide-1 analogue programmes exemplify efforts to enable needle-free metabolic therapies, though achieving reproducible bioavailability remains a primary technical hurdle. Collectively, these advances signal the continued expansion of nasal delivery as a versatile, systemic and CNS-drug delivery platform.
| Traditional Markets | Emerging Markets | Upcoming (New) Markets | |||
| Allergic rhinitis | Acute pain | Seizure rescue | Depression | Heart arrhythmia | Psychiatric agitation |
| Influenza vaccine | Opioid overdose | Migraine | Anaphylaxis | Traumatic brain injury | Metabolic disease |
Table 1: Traditional, emerging and upcoming (new) markets for liquid nasal drug products.
EARLY CMC AND DEVELOPMENT CONSIDERATIONS FOR LIQUID NASAL PRODUCTS
Early CMC strategy defines the technical and regulatory trajectory of a liquid nasal drug product. Unlike many conventional oral dosage forms, nasal products represent an integrated drug-device combination in which formulation properties, container-closure configuration and spray mechanics collectively determine clinical performance. Accordingly, early development should align formulation science, device platform selection, manufacturing strategy and analytical control within a unified target product profile.

Figure 1: Unit-dose nasal device (Nemera’s UniSpray).
Platform Selection
One of the earliest strategic decisions is selection of a unit-dose, bi-dose or multidose presentation. Unit-dose and bi-dose systems are generally preferred for emergency-use or high-potency products because they simplify microbial control and eliminate in-use contamination risk. These systems must demonstrate tight control of delivered volume (typically ± 10% or less, depending on product requirements), shot weight reproducibility and spray geometry consistency across production lots. An example of a unit-dose system is shown in Figure 1.
Nemera‘s UniSpray accurately delivers a single 100 µL dose in any orientation without priming. Multidose systems, which are more common for chronic indications, introduce additional CMC complexity, including preservative efficacy validation or implementation of preservative-free pump technologies that incorporate mechanical tip seals and one-way valve systems. Resyca’s Ultra Soft Nasal Pump, shown in Figure 2, uses Rayleigh spray technology to deliver a 45–70 µL fine, low-velocity plume, allowing for better nasal mucosal coverage and minimal dripping.3

Figure 2: (A) Multidose nasal device: Resyca Ultra Soft Nasal Pump Spray. (B) Image of nasal epithelium coverage due to low-velocity fine mist.3
Preservative-containing products require antimicrobial effectiveness testing (e.g. US Pharmacopeia <51>) and justification of preservative concentration relative to nasal tolerability and potential ciliary toxicity. Preservative-free multidose systems frequently require aseptic processing and validated container-closure integrity to maintain sterility assurance throughout labelled in-use periods.4
Manufacturability must be evaluated in parallel with device selection. Nasal products are particularly sensitive to fill volume accuracy, crimping or stopper placement (for unit-dose systems) and torque control (for multidose pumps). Variability in fill volume directly affects delivered dose and spray characteristics. Decisions regarding terminal sterilisation feasibility versus aseptic processing are often dictated by formulation stability and excipient compatibility. Aseptic filling introduces higher environmental classification requirements (ISO 5/7), media fill validation and sterility assurance considerations but may be necessary for preservative-free products or heat-sensitive actives. Early compatibility studies assessing sorption, leachables, silicone oil interactions and elastomer extractables are essential to prevent late-stage reformulation or device requalification.
Product Quality Control
Analytical development is equally critical. Regulatory guidance emphasises characterisation of both formulation and device performance attributes, including impurities, pH, viscosity, osmolality, microbial limits, delivered dose uniformity (DDU), droplet size distribution (e.g. laser diffraction Dv10/Dv50/Dv90), spray pattern and plume geometry.4 These attributes are directly linked to regional deposition within the nasal cavity and therefore to safety and efficacy. For suspension products, resuspendability, sedimentation rate and redispersibility must be quantified to ensure consistent dosing. Rheological profiling supports optimisation of mucosal residence time while minimising run-off and patient discomfort. Device-specific parameters – such as actuation force, stroke length and priming behaviour – should be incorporated into control strategies, particularly for combination products subject to human factors validation.
Early in vitro deposition testing using anatomical nasal cast models can provide predictive insight into regional deposition patterns, especially for programmes targeting the upper nasal cavity or potential nose-to-brain delivery.5 Such data may inform droplet size optimisation, plume angle adjustments or selection of breath-powered delivery technologies.
Collectively, these early CMC decisions establish the framework for clinical success and commercial scalability. Proactive integration of formulation, device engineering, manufacturing controls and analytical characterisation reduces downstream risk, shortens development timelines and supports a robust regulatory submission strategy.
FORMULATION CONSIDERATIONS FOR LIQUID NASAL PRODUCTS
Formulation development for liquid nasal products requires a careful balance of chemical stability, mucosal bioavailability, manufacturability and patient safety. Nasal formulations are inherently complex due to the combination of physicochemical properties, device interactions and local tolerability requirements. Early formulation decisions and excipient selection (Table 2) directly impact downstream manufacturing, analytical characterisation and clinical performance.
| Excipient Function | Excipient Name | In Commercial Product? | Commercial Product Example |
| Absorption enhancer | Dodecyl maltoside | Yes | Valtoco® |
| Alkylsaccharides (Intravail®) | Yes | Nayzilam® | |
| Chitosan | No | – | |
| Cyclodextrins (HPβCD) | Yes | Spravato® | |
| Bile salts (e.g. sodium deoxycholate) | No | – | |
| Mucoadhesive/residence enhancer | Chitosan | No | – |
| Carbomer (Carbopol) | Yes | XHANCE | |
| Hydroxypropyl methylcellulose (HPMC) | Yes | Flonase | |
| Sodium hyaluronate | Yes | Ryaltris | |
| Solubiliser (co-solvent) | Propylene glycol | Yes | Sprix® |
| Ethanol | Yes | Lazanda | |
| PEG 400 | Yes | Valtoco® | |
| Preservative | Benzalkonium chloride | Yes | Flonase |
| Phenylethyl alcohol | Yes | Nasonex | |
| Potassium sorbate | Yes | Patanase | |
| Buffer/pH adjuster | Product stability | Yes | Flonase |
| Weight loss | Yes | Zavzpret® | |
| Extractables and leachables | Yes | Spravato® | |
| Tonicity adjuster | Sodium chloride | Yes | Narcan® |
| Dextrose | Yes | Nayzilam® | |
| Mannitol | Yes | FluMistv® | |
| Antioxidant/stabiliser | EDTA (disodium edetate) | Yes | Flonase |
| Sodium metabisulfite | Yes | Neffy® | |
| Ascorbic acid | Yes | Afrin® | |
| Viscosity modifier | Microcrystalline cellulose + CMC (Avicel® RC) | Yes | Flonase |
| HPMC | Yes | Nasacort®Allergy 24HR | |
| Carbomers | Yes | XHANCE |
Table 2: Excipients used in liquid nasal drug products.
Formulation Type
Solution formulations are often the first approach considered for nasal delivery due to their simplicity, dose uniformity and compatibility with multidose and unit-dose devices. However, these systems demand rigorous control of solubility, pH, viscosity and chemical stability to maintain product integrity throughout shelf life. Solubilising agents (e.g. cyclodextrins, polyethylene glycol), buffering systems to maintain physiological pH (typically 4.5–6.5) and antioxidants (e.g. ascorbic acid, sodium metabisulfite) are commonly incorporated to mitigate degradation by hydrolysis or oxidation.
Viscosity plays a pivotal role in droplet formation, spray plume geometry and droplet size distribution; optimal viscosity ranges are typically 1–10 cP for aqueous solutions to ensure reproducible atomisation while minimising run-off and nasopharyngeal deposition variability.5
Suspension formulations present additional technical challenges. The particle size distribution of the API, typically achieved through micronisation, is critical for dose homogeneity, redispersibility and reproducible pharmacokinetics. Stabilising excipients – such as cellulose derivatives, xanthan gum or polysaccharides – are used to prevent sedimentation, creaming or particle agglomeration over the labelled shelf life. The interplay between suspension viscosity, pumpability and long-term stability often necessitates iterative optimisation, including compatibility testing with the intended delivery device to ensure consistent dose delivery across the container lifespan.1
Preservatives and Bioavailability
Preservative strategy is another crucial determinant, particularly for multidose systems. Conventional preservatives such as benzalkonium chloride, phenylethyl alcohol, or potassium sorbate are effective but may cause local irritation, ciliary toxicity or regulatory concerns for long-term use. Consequently, there is a growing trend towards preservative-free multidose devices, which rely on aseptic manufacturing and mechanical tip-seal pump designs to prevent microbial contamination. The choice of preservative – or the decision to omit one – directly influences extractables and leachables profiles, container-closure material selection and the overall microbial control strategy.4
Bioavailability-modifying excipients are critical for products requiring rapid systemic absorption or enhanced CNS exposure. Absorption enhancers (e.g. chitosan, alkylsaccharides), mucoadhesive polymers and solubilising agents can increase residence time, facilitate mucosal uptake and potentially support nose-to-brain delivery. Selection of these excipients must balance efficacy, nasal tolerability and regulatory acceptability. The integration of formulation design with device engineering, analytical characterisation and human factors considerations is essential to achieve reproducible delivery and therapeutic performance.
Ultimately, early formulation strategy must be informed by a combination of physicochemical profiling, stability studies, device compatibility testing and in vitro deposition modelling. These efforts ensure that liquid nasal products meet both regulatory and clinical requirements while maintaining manufacturability and patient safety.
CLINICAL AND COMMERCIAL PRODUCTION CONSIDERATIONS FOR LIQUID NASAL PRODUCTS
Transitioning a nasal formulation from development to clinical and commercial supply requires early alignment of scalable manufacturing processes, device integration and quality assurance strategies. Liquid nasal solutions and suspensions necessitate specialised filling technologies capable of accurately dispensing small volumes – often in the range of 50–200 µL per actuation – while maintaining dose uniformity and minimising product loss.
Commercial facilities commonly employ precision unit-dose filling systems, such as the Dara (Barcelona, Spain) SYX-E-UDS (Figure 3) and Groninger’s (Crailsheim, Germany) DFVN 1000, or multidose platforms such as the Filamatic (Baltimore, MD, US) ProLINE. These are available at Catalent’s nasal manufacturing site and incorporate high-accuracy metering, automated stopper placement and validated actuation mechanics to ensure consistent delivered dose and spray performance.

Figure 3: Unit-dose manufacturing line at Catalent: Dara SYX-E-UDS.
Scale-up and Validation
Scale-up from laboratory to commercial production introduces critical technical considerations. Formulation integrity must be preserved across larger batch sizes without altering rheological properties, droplet size distribution or suspension homogeneity. High-value or low-solubility APIs demand careful management of manufacturing losses during mixing, transfer and filling operations. Process analytical technologies (PAT), including in-line viscosity measurement, turbidity monitoring and near-infrared spectroscopy, can provide real-time quality assessment and support batch-to-batch consistency. Suspension products require rigorous control of particle size distribution throughout blending, holding and filling stages to prevent sedimentation or dose variability. Early generation of robust, scalable process parameters reduces the risk of late-stage reformulation or device requalification.
Validation and process characterisation are central to ensuring commercial readiness. Early identification of critical quality attributes – such as droplet size, plume geometry, DDU and microbial integrity – alongside critical process parameters for mixing, filling and device assembly, enables a risk-based validation strategy. Engineering and demonstration batches serve to confirm reproducibility, device-formulation compatibility and actuation performance under representative operating conditions. Validation programmes must encompass the entire drug-device combination, including assembly operations, container-closure integrity and, where applicable, aseptic or sterilisation processes to meet regulatory requirements.
“INTEGRATING FORMULATION SCIENCE, DEVICE ENGINEERING, ANALYTICAL CHARACTERISATION AND MANUFACTURING STRATEGY AT THE EARLIEST STAGES FOSTERS A COHESIVE,
HIGH-FIDELITY PRODUCTION PROCESS.”
Integrating formulation science, device engineering, analytical characterisation and manufacturing strategy at the earliest stages fosters a cohesive, high-fidelity production process. By proactively addressing scale-up, PAT implementation and validation design, developers can ensure that clinical supply is robust, reproducible and aligned with commercial demands, ultimately reducing development timelines and mitigating risk in the pathway to regulatory approval.
CONCLUSION
Liquid nasal dosage forms are experiencing unprecedented growth, with products ranging from small molecules to peptides and biologics, and encompassing solutions, suspensions, emergency therapies and nose-to-brain applications. The complexity of these modalities demands a fully integrated CMC strategy, where early alignment of formulation, device selection, analytical methods, manufacturing processes and microbial control ensures robust, reproducible and scalable outcomes.
Experience across the full development lifecycle is critical for translating innovative nasal therapies from concept to commercial supply. By combining advanced formulation science, precision device engineering, and rigorous manufacturing and analytical practices, developers can mitigate risk, accelerate timelines and deliver reproducible clinical performance. As the nasal therapeutic landscape continues to expand, such co-ordinated approaches will be central to enabling the next generation of high-value, patient-focused nasal drug products.
REFERENCES
- Costantino HR et al, “Intranasal delivery: Physicochemical and therapeutic aspects”. Int J Pharm, 2007, Vol 337(1–2), pp 1–24.
- Lochhead JJ, Thorne RG, “Intranasal delivery of biologics to the central nervous system”. Adv Drug Deliv Rev, 2012, Vol 64(7), pp 614–628.
- D’Angelo D et al, “Fluorescence-enabled evaluation of nasal tract deposition and coverage of pharmaceutical formulations in a silicone nasal cast using an innovative spray device”. J Adv Res, 2023, Vol 44, pp 227–232.
- “Guidance for Industry: Nasal Spray and Inhalation Solution, Suspension and Spray Drug Products-Chemistry, Manufacturing, and Controls Documentation”. US FDA, July 2002.
- Djupesland PG, “Nasal drug delivery devices: Characteristics and performance in a clinical perspective”. Drug Deliv Transl Res, 2013, Vol 3(1), pp 42–62.